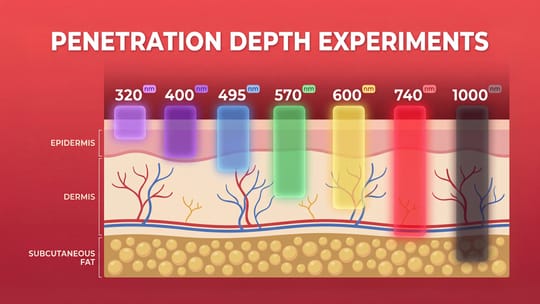
Photobiomodulation for the brain is getting serious scientific attention, especially for mild cognitive impairment (MCI) and early Alzheimer’s risk.
In this episode of Light Therapy Insiders, Bart Wolbers speaks with Dr. Neda Rashini, a physician-scientist focused on neurodegenerative research, about transcranial photobiomodulation, pilot trial results, mitochondrial biomarkers, inflammation, brain connectivity, and why this field may become an important part of future brain-health treatment plans.
Show Notes
In this episode, Dr. Neda Rashini explains how she went from being skeptical of photobiomodulation to leading clinical research on its use in people with mild cognitive impairment. The conversation covers why MCI matters, how placebo-controlled PBM trials are designed, what biomarkers were measured, and which outcomes looked promising in her pilot trial. They also discuss side effects, responder variability, and why bigger studies are still needed before stronger conclusions can be made.
Key Topics
Why Dr. Neda Rashini became interested in transcranial photobiomodulation
Although photobiomodulation has been around for decades, it has only recently gained traction in neuroscience research. Dr. Neda Rashini shares how her initial skepticism led her to investigate whether the biological claims behind PBM could actually be measured in clinical research.
Why mild cognitive impairment matters
MCI is often one of the earliest diagnosable stages before Alzheimer’s dementia develops. Dr. Neda Rashini explains why this stage may be a better window for intervention than later-stage disease, when neuronal loss may already be too advanced.
How the pilot trial was designed
The study compared an active transcranial photobiomodulation device against a sham device in people with MCI. Participants used what appeared to be the same headset, but only the active group received the full PBM treatment.
The biomarkers researchers measured
The trial looked beyond cognition alone. Dr. Neda Rashini’s team assessed mitochondrial biomarkers, inflammatory markers, functional brain connectivity, brain metabolites, and cognitive outcomes to better understand possible mechanisms.
What improved in the pilot study
The active group showed improved general cognition and recognition memory compared with the sham group. Researchers also observed changes in the lactate-to-pyruvate ratio, a reduction in IL-6 levels, and increased functional connectivity within the default mode network.
What did not significantly change
Not every marker moved. Some inflammation markers and neurodegeneration markers did not show significant differences, which Dr. Na says is important to report honestly — especially in a small pilot trial.
Why individual response matters
Some participants appeared to experience meaningful improvements in quality of life, while others noticed little change. This highlights the likely role of personalized treatment and the need to identify who responds best.
The next phase of the research
Dr. Na discusses the ongoing larger trial, which aims to recruit 60 participants, extend treatment from 6 to 8 weeks, and include follow-up testing after treatment ends.
How PBM could fit into Alzheimer’s care
Rather than viewing PBM as a complete solution, Dr. Na sees potential for it to become part of a broader treatment strategy — possibly alongside therapies that target amyloid and tau.
Side effects and safety considerations
While the treatment appears relatively accessible and low-risk overall, some participants reported headaches and scalp discomfort. Dr. Neda Rashini stresses that people with certain medical conditions, especially those at risk of bleeding or who use blood thinners, should be cautious and speak with a doctor first.
Resources Discussed
✨ The Vielight RX Gamma device used in the pilot trial was discussed as the transcranial photobiomodulation device studied in participants with mild cognitive impairment.
Grab a Vielight product here and use discount code ALEX10 for 10% off: https://aferg.co/vielight
🎧 Prefer to Listen?
Find Alex Fergus – Light Therapy Insiders on your favorite platform: